Researchers investigated the role of cyclodextrin derivatives in stabilizing carvedilol and chlorthalidone in a supersaturated state
Supersaturating poorly soluble drugs is a promising strategy to improve their bioavailability and oral adsorption. However, stabilizers are often necessary to maintain drug supersaturation and prevent crystallization. Recently, researchers explored the stabilizing effects of various cyclodextrin derivatives on the supersaturation of two model drugs, chlorthalidone and carvedilol, finding that methylated derivatives with higher hydrophobicity maintained drug supersaturation for longer periods. This study could help develop effective supersaturated drug formulations and facilitate their clinical use.
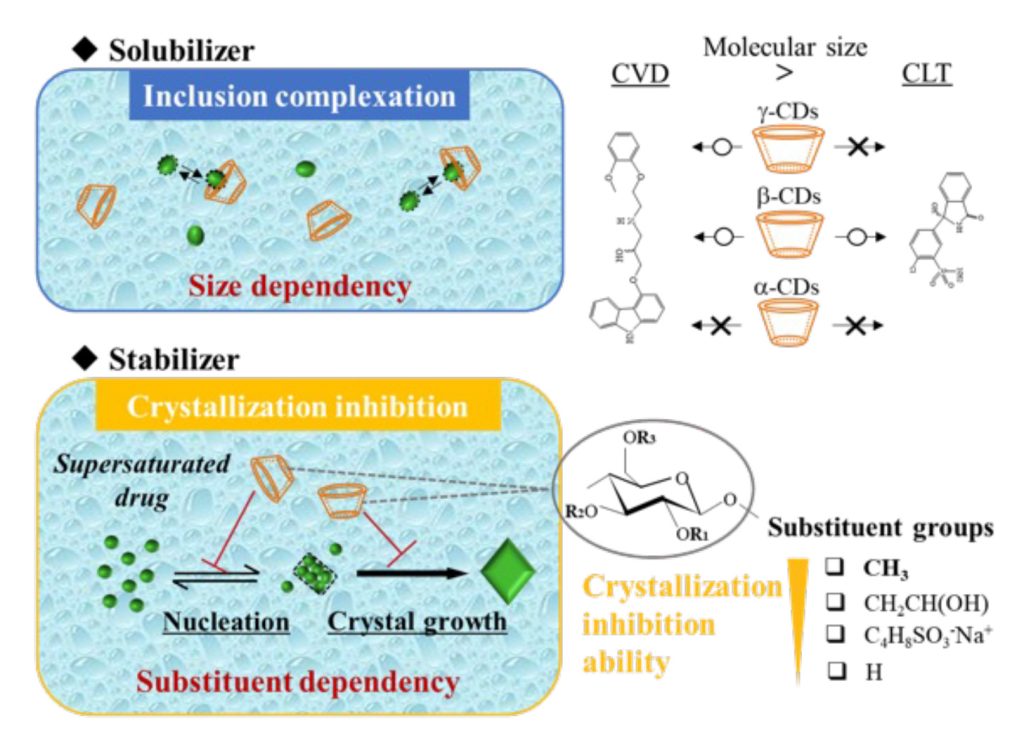
Title: The mechanism underlying the stabilization of supersaturated drugs with cyclodextrins.
Caption: Schematic illustration of inclusion complexation and crystallization inhibition abilities of cyclodextrins (CDs) on the model drugs: carvedilol (CVD) and chlorthalidone (CLT).
Credit: Kunikazu Moribe from Chiba University, Japan
License type: Original content
Usage restrictions: Cannot be reused without permission
In the medicine market, most newly introduced drugs and drug candidates show poor water solubility, which prevents their absorption in the body. This, in turn, limits their therapeutic efficiency. Solubilizing agents such as cyclodextrins (CDs) are commonly employed to enhance their solubility. CDs have a cyclic structure featuring a hydrophilic exterior and a hydrophobic cavity inside that can enclose drug molecules to form inclusion complexes. However, solubilization does not necessarily enhance drug adsorption in the body, since the solubilized drugs cannot readily pass through biological membranes.
One way to improve the permeability of drugs through biological membranes is by increasing their concentration in the solution to form a supersaturated state. However, supersaturated states are unstable as the drug tends to precipitate and form crystals, limiting its effectiveness. To prevent this, the addition of effective crystallization inhibitors is necessary to stabilize drug supersaturation over a long period. CD derivatives are particularly advantageous as they can solubilize drugs as well as inhibit their crystallization. However, the mechanism underlying their crystallization inhibition effect remains poorly understood.
In this light, a study led by researchers from Chiba University, Japan, recently investigated the impact of 12 different CD derivatives with varying hydrophobic cavity sizes on crystallization inhibition of two poorly water-soluble drugs – carvedilol (CVD) and chlorthalidone (CLT).
Their work was made available online on March 22, 2023, and published in Volume 637 of the International Journal of Pharmaceuticson April 25, 2023. It involved contributions from Professor Kunikazu Moribe, Associate Professor Kenjirou Higashi, and Assistant Professor Keisuke Ueda.
“Different solubilization abilities of CD derivatives lead to different drug supersaturation levels, resulting in a misestimation of their crystallization inhibition strength. Thus, a systematic analysis of the drug crystallization inhibition effect while considering the solubilization effect of CD derivatives is essential,” explains lead author Mengyao Liu, a Ph.D. student at the Laboratory of Pharmaceutical Technology at the Graduate School of Pharmaceutical Sciences at Chiba University.
The researchers evaluated the solubilization and crystal inhibition effect of each CD derivative on the two drugs by performing phase solubility tests and measuring the crystallization induction time – the time taken for a supersaturated solution to form crystals. In a phase solubility test, they dissolved the drugs in CD derivative solutions of different concentrations and examined their solubility. For measuring the crystallization induction time, the researchers added the drugs in drops to form supersaturated solutions and then analyzed their concentrations at different time intervals after removing the precipitated crystals.
The phase solubility tests revealed that the addition of β-CD and γ-CD derivatives improved the solubility of CVD. However, CLT’s solubility was enhanced only by the β-CD derivative. “Notably, the degree of solubility improvement depends on the ability to form stable inclusion complexes that, in turn, depends on the size fitness between the drug molecule and the CD cavity,” explains Prof. Moribe.
However, there was no correlation between the solubilization effect of the different CD derivations and their crystallization inhibition abilities. Moreover, methylated CD derivatives were more effective than their unmethylated counterparts in maintaining the supersaturated state. The researchers attributed the crystallization inhibition ability of the methylated CD derivatives to their highly hydrophobic outer surfaces, which sterically hinder the nucleation and crystal growth processes, and thus maintain the supersaturated state. “We have discovered a novel function of methylated CD derivatives as effective stabilizers for supersaturated drugs, an effect that is independent of their inclusion complex formation ability,” highlights Prof. Moribe.
In conclusion, these findings shed light on the potential application of CD derivatives in drug-supersaturated formulations, which, in turn, could facilitate the clinical use of poorly water-soluble drug candidates.
About Professor Kunikazu Moribe
Dr. Kunikazu Moribe is a Professor at the Laboratory of Pharmaceutical Technology at the Graduate School of Pharmaceutical Sciences at Chiba University in Japan. His research focuses on physical pharmacy, pharmaceutical technology, and drug delivery systems. He has published over 183 papers on pharmaceutics, which have been cited over 3,000 times. In recognition of his contributions to the field, he was conferred the Asahi Kasei Pharmaceutical Technology Research Award for Young Scientists in 2013 by the Academy of Pharmaceutical Sciences and Technology Japan.
Reference:
Authors: Mengyao Liu 1, Kenjirou Higashi 1, Keisuke Ueda1, Kunikazu Moribe1
Title of original paper: Supersaturation maintenance of carvedilol and chlorthalidone by
cyclodextrin derivatives: Pronounced crystallization inhibition ability of methylated cyclodextrin
Journal: International Journal of Pharmaceutics
DOI: 10.1016/j.ijpharm.2023.122876
Affiliations: 1Graduate School of Pharmaceutical Sciences, Chiba University
Contact:
Kunikazu Moribe
Laboratory of Pharmaceutical Technology, Graduate School of Pharmaceutical Sciences, Chiba University
Address: 1-8-1, Inohana, Chuo-ku, Chiba, 260-8675, JAPAN
Email: moribe@faculty.chiba-u.jp
Public Relations Office, Chiba University
Address: 1-33 Yayoicho, Inage, Chiba 263-8522, Japan
Email: koho-press@chiba-u.jp
Tel: +81-43-290-2018





